Le plan France Médecine Génomique (PFMG)
Depuis 2016, la France s’est dotée d’un plan national, le plan France Médecine Génomique 2025 (PFMG) visant à intégrer le séquençage du génome complet au parcours de soins des patients et disposant d’un fort potentiel pour développer la recherche ainsi qu’une filière nationale de la médecine génomique.
Le PFMG contribue à la mise en œuvre d’un accès équitable à la médecine génomique sur l’ensemble du territoire et à l’organisation d’un nouveau continuum soin-recherche, en s’appuyant sur 3 piliers indissociables
:
- Un réseau de laboratoires de séquençage à très haut débit qui produit les données génomiques et les analyse pour les délivrer dans le cadre du parcours de soin ;
- Un centre d’analyse des données (le Collecteur Analyseur de Données) visant à permettre la réanalyse des données par les différents utilisateurs dans le cadre du soin mais aussi leur réutilisation dans de très nombreux domaines de la recherche ;
- Un centre d’innovation et d’expertise (CRefIX).
Les deux premiers laboratoires de séquençage à très haut débit,
AURAGEN et
SeqOIA, couvrent l’ensemble du territoire.
À ce jour, il est possible de prescrire un séquençage à très haut débit pour
61 pré-indications :
- 51 concernent les maladies rares
- 10 concernent les cancers et l’oncogénétique
Elles ont été retenues à l’issue d’une phase de validation effectuée par un groupe de travail piloté par la Haute Autorité de Santé.
Le PFMG s’organise autour du patient, depuis la prescription médicale d’un examen de séquençage du génome jusqu’à la restitution des résultats par le médecin prescripteur. Un parcours de soins générique a été défini et adapté aux spécificités de chacune des 61 pré-indications :
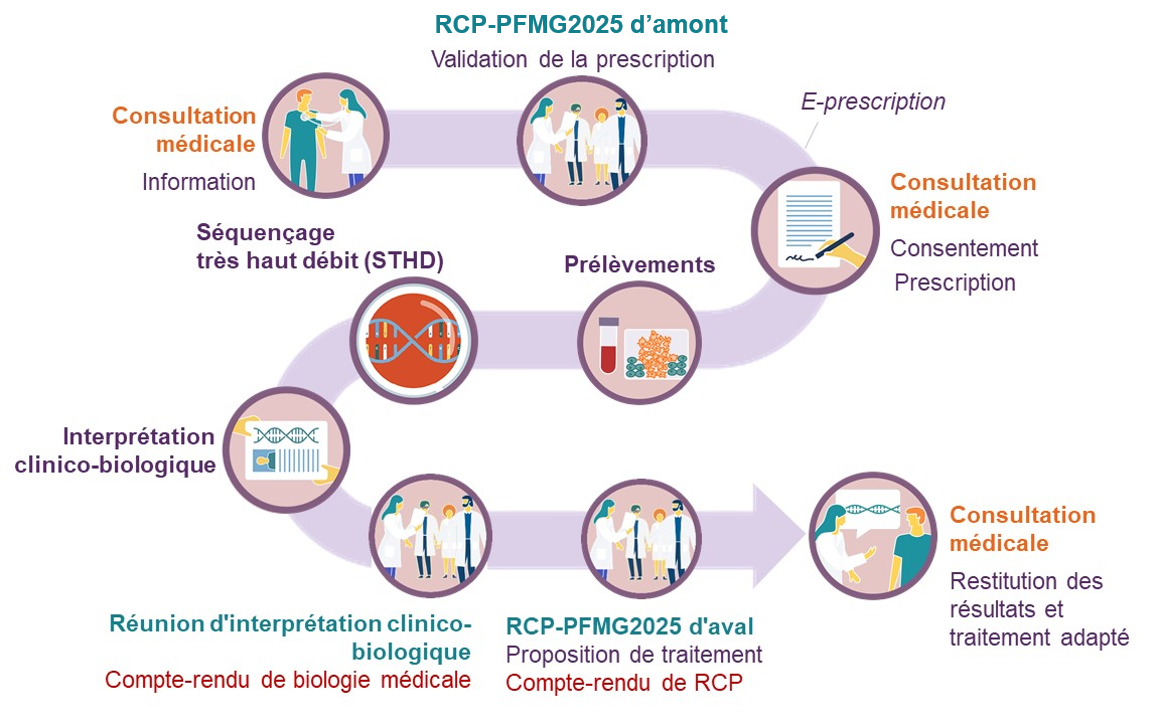
Pour sa mise en œuvre, le plan France médecine génomique 2025 intègre également des actions transversales de support, rassemblant de nombreux acteurs issus d’organismes de recherche, d’universités, d’établissements de santé, de ministères, d’agences sanitaires et des représentants de patients :
Aujourd’hui, les deux premiers laboratoires de séquençage, AURAGEN, et SeqOIA, le CRefIX et les circuits de prescription sont opérationnels pour la grande majorité des 61 pré-indications dans le champ des maladies rares et des cancers.

En fin d’année 2020, 2855 prescriptions ont été validées en RCP-FMG d’amont :
- 2176 pour les maladies rares
- 86 pour l’oncogénétique
- 593 pour les cancers
1880 dossiers complets ont été reçus, dont 1279 dossiers ont été séquencés. Des comptes rendus ont été signés pour 214 d’entre eux, dont 81 pour les maladies rares et 133 pour les cancers.
Le travail réalisé par le PFMG au cours des deux dernières années pour organiser les parcours de soins dans le cadre d’une soixantaine de préindications se traduit par une très bonne dynamique de participation de patients au deuxième semestre 2020 (+97%), à la fois pour les maladies rares (+84%) et les cancers (+167%).
Les circuits en amont du séquençage à très haut débit sont organisés en lien avec les filières de santé maladies rares et les acteurs de la cancérologie. Les circuits en aval du séquençage à très haut débit sont aujourd’hui au cœur du travail du PFMG.
Déploiement des chargés de parcours génomique sur le territoire
Le Plan National Maladies Rares 3 a financé le déploiement de 24 chargé.e.s de parcours génomique sur l’ensemble du territoire français afin de fluidifier le parcours génomique dans le champ des maladies rares. À ce jour, 11 chargé.e.s de parcours génomique sont recruté.e.s et 13 en cours de recrutement. Ils/elles ont pour mission de former et accompagner les prescripteurs à utiliser les logiciels de e-prescription SPICE et HYGEN, suivre et accompagner des RCP-FMG, et, de façon opérationnelle, faciliter les prescriptions (information au patient et à sa famille, signature du consentement, logistique d’envoi des prélèvement). La liste des chargé.e.s de parcours génomique et leur périmètre géographique d’activité est consultable sur le site internet en cliquant ici.

La collaboration étroite entre le Plan National Maladies Rares 3 (PNMR3) et le Plan France Médecine Génomique (PFMG) a permis de mettre en place une mesure permettant de faciliter les prescriptions et fluidifier les circuits en amont du séquençage des examens pangénomiques, Le PNMR3 finance 24 postes de chargés de parcours génomique déployés sur 21 sites hospitaliers différents, sièges de plateforme d’expertise maladies rares et ou de CRMR/CCMR, et sur l’ensemble du territoire français et des DROM. Pour une mise en place optimale du PFMG, le recrutement de chargés de parcours génomique est également envisagé pour les pré-indications dans le champ des cancers.
Quelles sont les missions du chargé de parcours génomique ?
1. Assister les cliniciens CRMR/CCMR qui prescrivent des séquençages de génome sur AURAGEN et SeqOIA dans le cadre des préindications maladies rares :
- Préparation des dossiers pour présentation aux Réunions de Concertation Pluridisciplinaire dédiées (RCP-FMG d’amont), avec notamment remplissage des fiches de RCP dédiées et du bilan de suivi des préindications;
- Participation aux RCP-FMG d’amont avec rédaction des comptes-rendus de RCP si besoin ;
- Saisie des dossiers de patients dans les outils de e-prescription en ligne (SPICE pour la plateforme SeqOIA et HYGEN pour la plateforme AURAGEN) ;
- Aide à la création des comptes prescripteurs dans les outils de e-prescription en ligne pour les cliniciens CRMR/CCMR ;
- Et saisie des indicateurs individuels (par patient) d’évaluation médico-économique des préindications dans l’outil dédié.
2. Pour les chargés de parcours génomique avec un diplôme de conseiller en génétique : consultations autonomes, sous la responsabilité d’un médecin généticien, pour informer les patients et recueillir leur du consentement éclairé.
3. Formation des cliniciens prescripteurs aux outils de prescription en ligne (SPICE pour le laboratoire SeqOIA et HYGEN pour le laboratoire AURAGEN) ;
4. Assistance à la mise en place du circuit des prélèvements sanguins jusqu’au lieu d’enlèvement par le transporteur dédié ;
5. Collaboration avec les équipes d’AURAGEN et SeqOIA pour faciliter les circuits et modalités de prescription.